4 vinylphenol exists in all eukaryotes ranging from yeast to humans.
Vinyl carbon nmr.
Carbon 13 c13 nuclear magnetic resonance most commonly known as carbon 13 nmr or 13 c nmr or sometimes simply referred to as carbon nmr is the application of nuclear magnetic resonance nmr spectroscopy to carbon it is analogous to proton nmr 1 h nmr and allows the identification of carbon atoms in an organic molecule just as proton nmr identifies hydrogen atoms.
Vinyl acetate is used to make other industrial chemicals.
This is also known as 3 buten 2 one amongst many other things here is the structure for the compound.
The nmr parameters are compared for the entire series of.
Substituents can move the resonance out of the listed range esters amides acids ketones aldehydes.
The key difference between these two structural components is the number of carbon and hydrogen atoms.
The c 13 h couplings were also obtained for the other chloroethylenes.
Second order effect like ab abx aa xx can be simulated as well.
Both groups own a double bond between two carbon atoms where all the other atoms are bonded through single bonds.
Simulate and predict nmr spectra directly from your webbrowser using standard html5.
Vinyl aromatics nitriles rocr 3 arcr 2 h alkyne r 3coh o 1h 2nmr shift ranges δ ppm vinyl r 3c f r 3c clrc i r 3c br rccr 3 o δ ppm 13c nmr shift ranges r 2nh r 2ncr h approximate nmr shift ranges note.
4 vinylphenol exists as a solid slightly soluble in water and a very weakly acidic compound based on its pka.
Key difference allyl vs vinyl both allyl and vinyl groups have slightly similar structures with a small variation.
Carbonic acid cyclic vinylene ester.
Vinyl chloride is primarily used to make polyvinyl chloride to manufacture plastics.
4 vinylphenol participates in a.
It is a clear colorless liquid with a sweet fruity smell.
4 vinylphenol also known as p hydroxystyrene belongs to the class of organic compounds known as styrenes.
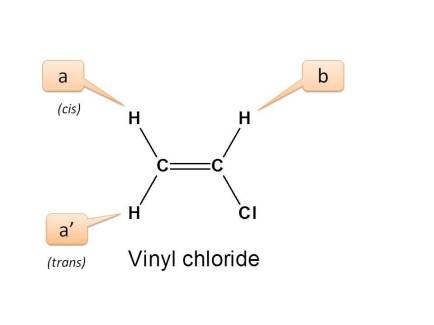
Three deuterated species of vinyl chloride were used to facilitate the analysis and to obtain all the c 13 h couplings in this molecule.
You can also simulate 13c 1h as well as 2d spectra like cosy hsqc hmbc.
Vinyl chloride is a chlorinated hydrocarbon occurring as a colorless highly flammable gas with a mild sweet odor that may emit toxic fumes of carbon dioxide carbon monoxide hydrogen chloride and phosgene when heated to decomposition.
The c 13 nmr spectrum for but 3 en 2 one.
Allyl groups have three carbon atoms and five hydrogen atoms.
These are typical chemical shifts.
It is very flammable and may be ignited by heat sparks or flames.
Predict 13c carbon nmr spectra directly from your webbrowser using standard html5.
Vinyl acetate is an industrial chemical that is produced in large amounts in the united states.
These are organic compounds containing an ethenylbenzene moiety.
The two peaks at 137 and 129 are due to the carbons.